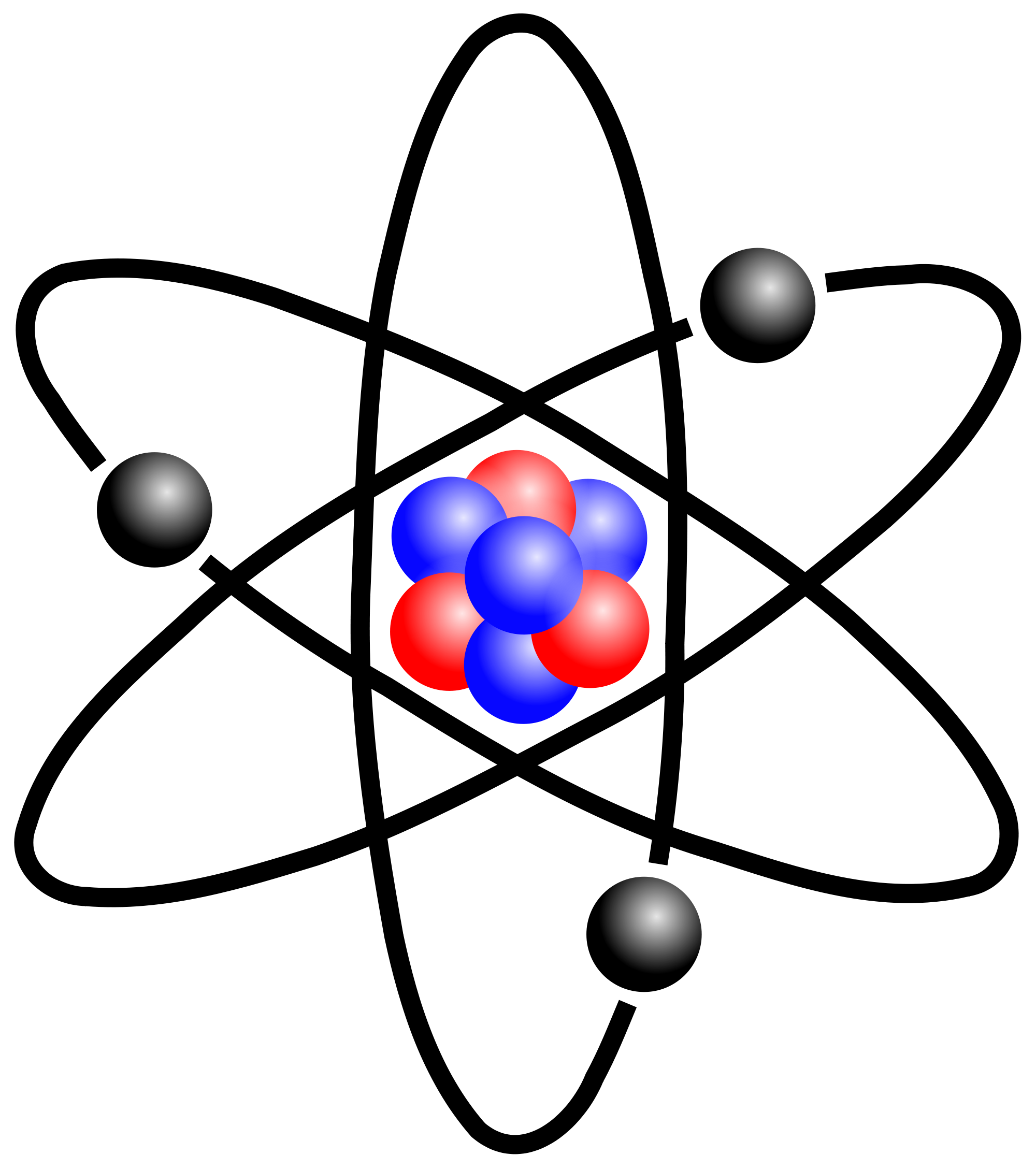
And so we can go ahead and, uh, indicate that a Here is the correct answer to this example problem and that concludes this example. And so we can pretty much say that their mass is about equal to zero. And so that can remind us that electrons are negatively charged. For example, hydrogen atoms in alkenes absorb energy in the 56 region. And so electrons are negatively charged and recall that electrons kind of sounds like getting electrocuted and getting electrocuted is something negative. The hydrogen atom (H1) attached to the silicon center could be located in the. And, of course, this Onley leaves the electrons, which is the correct answer for this example problem. Molecules are held together by shared electron pairs, or covalent bonds. The usual charge of an element is common to its group. So once again we can eliminate nutrients. There are four ways to find the charge of an element: Use the periodic table. Taking cobalt as an example, we further produce a series of Co single-atom catalysts with varying loadings to investigate the influence of density on regulating the electronic structure and. And so because protons are positively charged, they're not going to be negatively charged, and so we can eliminate, answer, option beat and also recall that neutrons are, as their name implies, new troll and neutral means that it has a net charge of zero, and so it will not be negatively charged. So now we're between either electrons, protons or neutrons, which are the three sub atomic particles of a typical Adam recall that the P and protons can remind us that this subatomic particle is positively charged so you can think the P and protons is for the P and positively charged. For example, any atom with an atomic number of 8 (its nucleus contains 8 protons) is an oxygen atom, and any atom with a different number of protons would be a different element. But because we have not yet introduced these two terms, we should have been able to figure out that these two are not going to be the correct answer for this problem. An atom can be classified as a particular element based solely on its atomic number. 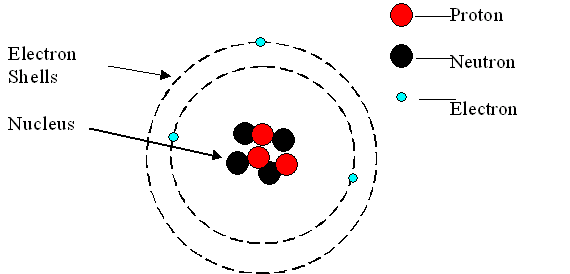
However, later in our course we definitely will talk about these two terms. Now, so far, at this point in our course, we have not yet introduced ions or polymers. For example, table salt is made up of two kinds of atoms like Sodium and. And it says that negatively charged particles of Adams with almost no mass are called either electrons, protons, neutrons, ions or polymers. Ans: The easiest definition of an atom is 'The smallest uni. So here we have an example problem that wants us to fill in the sentence here, using one of these five potential answer options down below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
